|
||
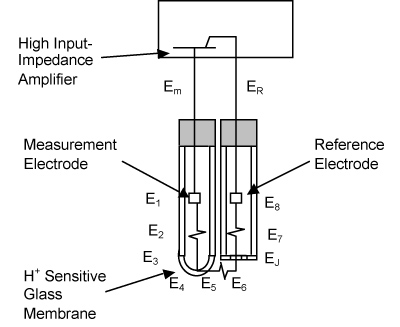
E-Zine June 2013The figure below shows the equivalent circuit for glass pH analyzer where the electrodes are solid conductors used to establish contact with nonmetallic parts of the circuit.
The measurement electrode is a silver wire coated with silver chloride (AgCl) that is used to detect the electrical potential between itself and the chloride-containing buffer solution that surrounds it. The buffer solution is formulated to maintain a pH of 7 and is prevented from contacting the process fluid by a glass body (on its sides) and a special glass membrane on the bottom. This glass membrane generates an electrical potential both on its inside and outside surfaces that is related to the concentration of hydrogen ions in the adjacent solution. The reference electrode is used to generate a stable electrical potential regardless of the concentration of the chemical species in the sample while also providing a path for electrical continuity to the measurement electrode.
Excerpted from The Consumer Guide to Industrial pH and ORP Instrumentation ISSN 1538-5280 |
||