|
||
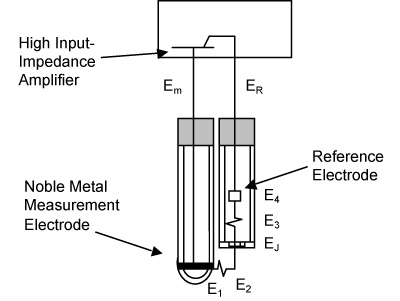
E-Zine October 2013Oxidation-reduction potential (ORP) measurement half-cells consist of a single noble metal electrode that is typically simpler than their pH counterparts. Noble metals such as gold and platinum are used because they do not react with most aqueous solutions and neither dissolve nor form oxide layers that could reduce contact between the metal and ions in solution. ORP measures the electrical potential created in an aqueous solution between the noble metal electrode and a reference half-cell. As with pH measurements, the reference half-cell maintains the same potential regardless of the solution in which it is immersed. An equivalent circuit diagram for an ORP measurement system is shown below.
Whereas pH measurements generate an electrical potential that is a linear function of the logarithm of one concentration, ORP measurements generate an electrical potential that is a logarithmic function of the ratio of two concentrations. In many applications there can be three or more concentrations involved in the oxidation-reduction reactions. For this reason, ORP measurement is often a poor choice for monitoring or controlling the concentration of a single species. In process applications, ORP measurement is typically better suited for monitoring or alarming overall process changes that would then trigger manual intervention to determine the exact cause of the ORP disturbance. It is also important to note that in many complex applications, hydrogen ions are involved in the oxidation-reduction reactions so the hydrogen ion concentration also appears in the logarithmic term and causes an interaction between the pH and ORP measurements.
Excerpted from The Consumer Guide to Industrial pH and ORP Instrumentation ISSN 1538-5280 |
||